In an era when drug safety decisions must be made in real time, automated case intake is essential as manual processing of Individual Case Safety Reports (ICSRs) has become a costly bottleneck. With rising adverse event volumes, unstructured data sources, and stringent compliance mandates, relying on outdated workflows isn’t just inefficient; it’s a liability.
The Hidden Cost of Manual ICSR Case Intake in Pharmacovigilance
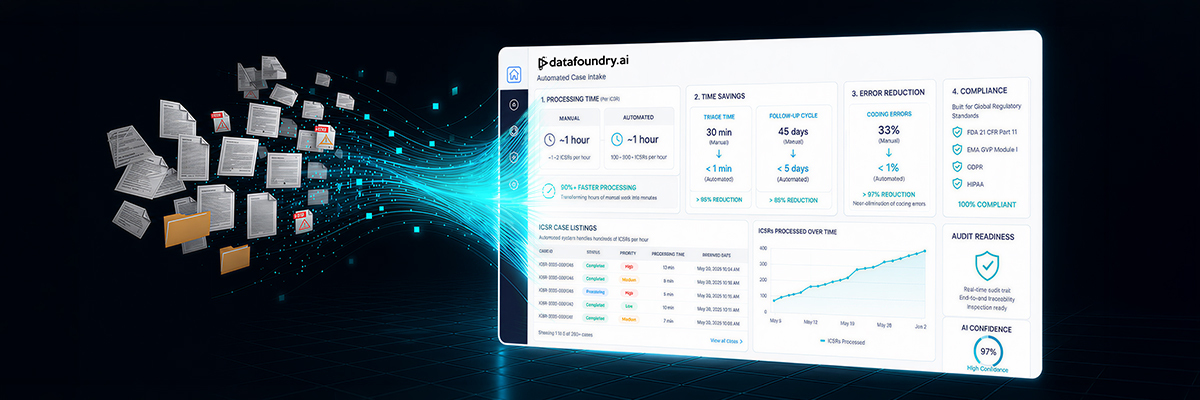
Manual case intake is a time-consuming process, often requiring 15–40 minutes to process a simple ICSR and up to four hours for complex adverse event cases. These figures do not yet account for the additional burden of peer reviews, medical coding, compliance checks, or regulatory paperwork. As a result, safety teams find themselves overwhelmed, buried in documentation, racing to meet deadlines, and struggling to maintain quality.
The Risks of Manual Oversight and Incomplete Data
Manual case intake not only slows workflows but also compromises data quality. When information comes in from scattered sources like emails, PDFs, or even social media posts, consistency often suffers. Nearly one-third of manually processed reports contain coding errors, while another 8% misclassify critical adverse events, for instance, labeling drug abuse as a simple overdose. The consequences of such errors are significant. A study published in the Journal of Clinical Oncology found that only 14% of manually reviewed reports contributed to actionable safety decisions, while the remainder contributed minimal insights beyond background noise. These aren’t just operational inefficiencies; they’re blind spots that can derail timely regulatory action.
How Automated Case Intake Changes the Pharmacovigilance Workflow
Fortunately, the future of case intake need not adhere to outdated workflows. Automated Case Intake (ACI) systems are purpose-built to address precisely these inefficiencies, accelerating case processing, improving accuracy, and ensuring compliance at scale. Unlike manual workflows that rely on human effort and subjective triage, ACI platforms use AI and rules-based engines to classify, route, and validate safety data in real time. This approach leads to faster throughput, significantly fewer errors, and a safety function that scales in alignment with demand.
Automation leads to significant operational efficiency. For instance, while a human reviewer can typically process just over one ICSR per hour, automated platforms can handle hundreds in the same timeframe. Triage times, which take 30 minutes per case manually, are cut down to under a minute, and follow-up cycles shrink from 45 days to less than 5. For safety teams under pressure to move fast without compromising accuracy, the performance gap between manual and automated workflows is simply too substantial to overlook.
Navigating the ICSR Data Landscape with AI-Driven Case Intake
Individual Case Safety Reports (ICSRs) flow into pharmacovigilance teams from a remarkably diverse ecosystem of both structured and unstructured sources, each carrying pieces of the safety puzzle. On the structured side, standardized electronic formats such as the ICH E2B(R3) specification form the backbone, with dedicated portals like FDA’s MedWatch, clinical trial databases, disease registries, and electronic health records (EHR) systems capturing patient demographics, suspect drug information, adverse event fields, and reporter credentials in predefined fields. However, the complete narrative often resides within unstructured data sources, free-text narrative descriptions within ICSR forms; voice-recorded hotline calls and patient-support conversations; detailed emails from clinicians, patients, or industry partners; and even posts on social media and forums, where people describe side effects using slang, emojis, or off-hand comments. Add to that electronic health record clinical notes, discharge summaries, medical literature case reports, and patient-support program dialogues, thus we have a wealth of contextual detail with an appropriate paper-trail.
Upon reception of these varied data inputs, AI takes the helm by harmonizing and enriching every data point into a uniform safety dataset. Natural Language Processing (NLP) engines can scan free-text reports, clinical notes, transcribed call-center recordings, emails, and social posts to extract core details such as patient demographics, suspect drugs, adverse event descriptions, and timing, all in real time. Optical Character Recognition (OCR) can play a pivotal role in digitizing handwritten or scanned forms, while algorithms such as Named Entity Recognition (NER) can populate structured fields without manual intervention. Transformer-based deep-learning models can be used to map extracted terms to standardized coding systems like MedDRA, adeptly handling multilingual content, medical abbreviations, and synonymous phrases. Duplicate-detection algorithms can be utilized to merge duplicate reports, and voice-to-text systems convert audio submissions into machine-readable text. Finally, AI-powered Bayesian networks and survival-analysis models evaluate causality and dose-response trends across all sources. The outcome: over 90% faster processing than manual methods, complete audit trails, and a consistently compliant safety dataset, no matter where the information originated.
Datafoundry’s Automated Case Intake Solution for Pharmacovigilance
Datafoundry’s Automated Case Intake (ACI) solution flips the script on manual workflows. ACI changes intake from a labor-intensive process into an intelligent, real-time workflow by immediately applying pharmacovigilance-tuned NLP and rules engines to reports. It automatically extracts key details, flags urgent cases for immediate attention, and feeds downstream workflows with standardized, audit-ready data, so the Safety experts can dive straight into evaluating risk and safeguarding patients.
Smarter Extraction, Faster Action
At its core is a powerful natural language processing engine trained specifically in pharmacovigilance language and ICSR taxonomies. The tool identifies and extracts critical fields such as adverse event terms, patient demographics, suspect drugs, dates, and reporter details with contextual accuracy. That means far fewer missed data points and significantly reduced manual cleanup.
Intelligent Routing That Adapts to Risk
Once data is captured, ACI automatically prioritizes, and routes cases based on severity, geographic region, regulatory timelines, and product-specific rules. High-risk cases are fast-tracked to medical review teams, while routine reports are categorized and queued for standard processing, all with complete, transparent audit trails.
Built for Scale, Designed for Compliance and Security
As global case volumes rise, ACI is built to scale through cloud-native architecture and seamless integration with existing pharmacovigilance systems. The platform is also designed with security at its core, employing encrypted data pipelines, access controls, and continuous alignment with HIPAA, GDPR, FDA, and EMA regulations. Each case action is time-stamped, auditable, and fully compliant with international data protection standards.
Intake as a Strategic Lever
Traditionally viewed as a tactical, low-value task, intake can be your strategic advantage. By converting a bottleneck into an intelligent entry point, Datafoundry’s ACI tool lays the foundation for end-to-end automation, improving signal detection, accelerating case processing, and delivering the data quality needed for confident, compliant decision-making.
By embracing Datafoundry’s Automated Case Intake Tool, teams unlock faster processing, near-elimination of preventable errors, and a scalable, audit-ready platform built for the demands of modern drug safety. In pharmacovigilance, every second, and every data point, matters. Automation isn’t just the next step; it’s the foundation for protecting patients and advancing the future of healthcare.
Frequently Asked Questions About Automated Case Intake in Pharmacovigilance
1. What is Automated Case Intake (ACI)?
ACI is an AI-powered system that automates the intake, processing, and classification of Individual Case Safety Reports (ICSRs), improving speed, accuracy, and compliance.
2. Why is manual case intake inefficient?
Manual processing is time-consuming, error-prone, and difficult to scale, often taking up to hours per case and leading to inconsistent data quality.
3. How does ACI improve processing speed?
ACI reduces case processing time from hours to minutes by automating data extraction, triage, and routing in real time.
4. What types of data can ACI handle?
ACI can process both structured data (e.g., EHRs, E2B formats) and unstructured data (e.g., emails, PDFs, social media, voice recordings).
5. What technologies power ACI systems?
Key technologies include Natural Language Processing (NLP), Optical Character Recognition (OCR), Named Entity Recognition (NER), and machine learning models.
6. How does ACI improve data quality?
It standardizes and validates incoming data, reduces coding errors, detects duplicates, and ensures consistent classification of adverse events.
7. Is ACI compliant with regulatory requirements?
Yes, ACI systems are designed to align with global regulations such as HIPAA, GDPR, FDA, and EMA, with full audit trails and secure data handling.
8. How does ACI support pharmacovigilance teams?
It automates repetitive tasks, prioritizes high-risk cases, and enables teams to focus on analysis and decision-making rather than manual data entry.
9. Can ACI scale with increasing case volumes?
Yes, cloud-based ACI platforms are built to handle large volumes of reports efficiently without compromising performance.
10. Why is ACI considered a strategic advantage?
It transforms intake from a bottleneck into an intelligent entry point, enabling faster insights, better signal detection, and improved patient safety outcomes.
Author: Ryanka Chauhan