
The pharmaceutical industry is advancing at an unprecedented pace, with new drugs and therapies continually emerging to improve patient outcomes. Yet, the critical focus on patient safety has never been more intense. For small and medium-sized pharmaceutical companies, this presents both an opportunity and a challenge. The opportunity lies in delivering innovative and safe medicines to the market, while the challenge is to ensure that safety practices are on par with the industry giants. This is where drug safety automation, particularly with the likes of DF Safety AI, can transform the landscape. In this blog, we will delve into the world of drug safety automation and explore how it can empower small and medium pharma companies in the realms of people, process, and system.
A Paradigm Shift in Drug Safety
Traditionally, drug safety has been a complex and often labor-intensive process that involves the collection, assessment, and reporting of adverse events. With the increasing volume of data and stringent regulatory requirements, the demand for accuracy and efficiency has escalated.
For small and medium pharma, this can pose substantial challenges:
Limited Resources: Smaller companies often operate with more constrained resources, making it difficult to compete in terms of data processing and safety assessments.
Regulatory Compliance: Regulatory authorities hold every pharmaceutical company to the same high standards, regardless of their size. Compliance is non-negotiable, but the cost of compliance can be substantial.
Data Management: The sheer volume of data generated in today’s pharmaceutical environment can be overwhelming, and handling this data efficiently can be a significant undertaking.
DF Safety AI: Empowering Small and Medium Pharma
This is where DF Safety AI emerges as a game-changer. It is an integrated solution that combines automation, advanced analytics, and vigilant monitoring to streamline drug safety operations for pharma companies, irrespective of their size.
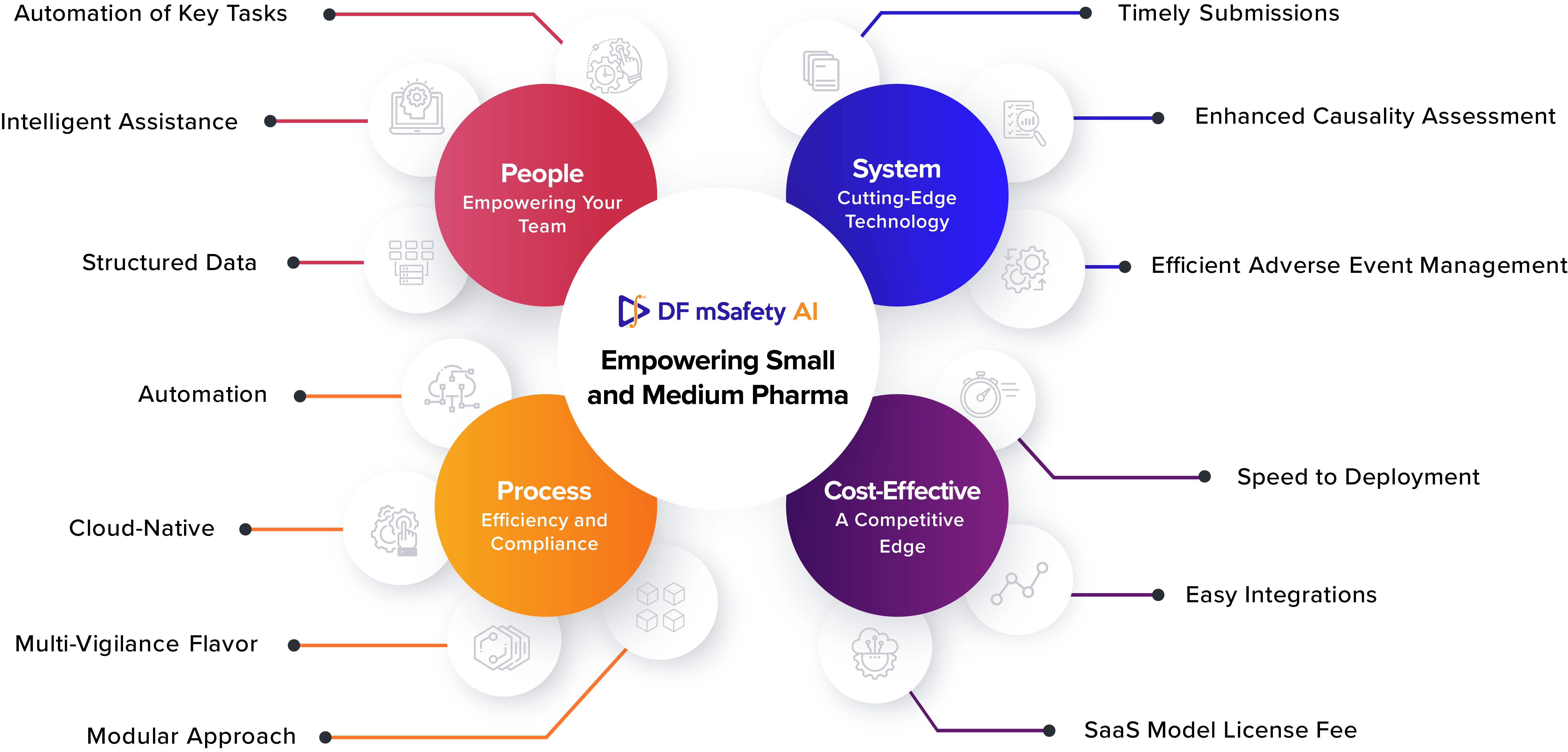
1. People: Empowering Your Team
DF Safety AI is designed with your team in mind. It simplifies the tasks associated with safety case processing, causality assessment, and risk management. Your staff can focus their expertise where it matters most, making better use of their time and skills.
Automation of Key Tasks: The solution automates many time-consuming processes, reducing manual effort.
Intelligent Assistance: With AI-driven automation and integrated approaches, causality assessments become more accurate and reliable.
Structured Data: The solution provides structured, standardized data, facilitating automation and informed decision-making.
2. Process: Efficiency and Compliance
Efficient processes are essential for smaller pharmaceutical companies to compete on a global stage. DF Safety AI enhances your processes by ensuring that safety cases are processed accurately and on time.
Timely Submissions: The solution streamlines processes to ensure submissions are made promptly, reducing the risk of non-compliance.
Enhanced Causality Assessment: DF Safety AI’s intelligent automation improves the accuracy of causality assessments, resulting in safer drug management.
Efficient Adverse Event Management: By automating critical tasks, the solution reduces costs, improves efficiency, and ensures comprehensive adverse event management.
3. System: Cutting-Edge Technology
DF Safety AI is built to meet the challenges of modern drug safety, offering features that are essential for both small and medium-sized pharma companies.
Automation: The solution automates key process steps, from case intake to causality assessment, significantly improving efficiency.
Cloud-Native: Unlike older solutions, DF Safety AI is cloud-native, offering flexibility and scalability. This ensures you can grow without the limitations of traditional systems.
Multi-Vigilance Flavor: The solution allows you to switch between various vigilance areas, including pharma, cosmetics, vaccines, and medical devices, all within the same instance.
Modular Approach: Whether you need a full safety database solution or a specific module, DF Safety AI offers flexibility.
4. Cost-Effective: A Competitive Edge
The journey to safer drugs and regulatory compliance is often accompanied by concerns about costs. However, DF Safety AI is designed with the cost-effectiveness of small and medium pharma companies in mind. It offers:
Speed to Deployment: Traditional drug safety systems can take months, if not years, to implement. In contrast, DF Safety AI can be up and running in less than a few weeks for small and medium-sized companies reducing the time and resources required for setup and training.
Easy Integrations: DF Safety AI seamlessly integrates with existing systems and databases, saving costs associated with complex overhauls.
SaaS Model License Fee: The platform offers cost-effective Software as a Service (SaaS) model, eliminating the need for substantial upfront investments.
Embrace the Future of Drug Safety
The pharmaceutical landscape is evolving rapidly, and small and medium pharma companies need to keep pace. Automation, like that offered by DF Safety AI, is not just a luxury; it is a necessity for ensuring both patient safety and regulatory compliance. As small and medium-sized pharma companies continue to play an essential role in the industry’s progress, it is crucial to empower them with tools that can match the capabilities of their more extensive counterparts. DF Safety AI offers the potential for a safer, more efficient, and more agile future in drug safety, ensuring that companies of all sizes can contribute to the betterment of global health outcomes.
Authors: Ryanka Chauhan – PM, Safety & Nandit Suresh – PM, Safety